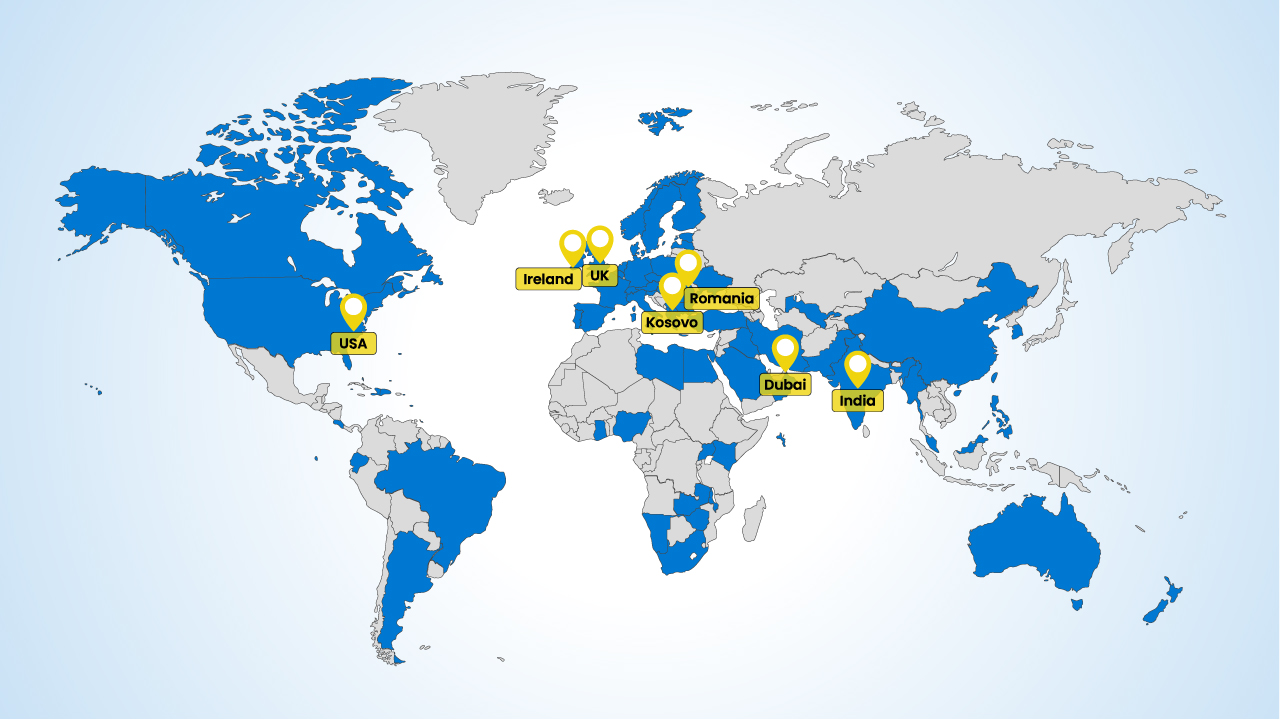


We use our fully owned and controlled strategic supply hubs in the EU, UK, UAE and India to provide quick, accurate supply, everywhere.
Our fully licenced, high quality pharmaceutical warehousing is equipped to handle all types of pharmaceuticals, temperature ranges and medical devices. With in-house relabelling capability, we can quickly and effectively meet any labelling requirements.
We handle the entire supply chain, monitoring shipments 24-7 from our supply chain control tower. We constantly monitor security and integrity, including temperature, humidity and GPS location. We also manage the entire import and customs clearance process.
Our in-house pharmacy also allows you to add dispensing and direct-to-patient delivery to your core program.
Our global network of GxP registered warehousing and distribution sites are located in strategic locations to allow a global network. We mitigate disturbance risk to the supply chain by holding stock in differing locations. They are linked to our global inventory and quality systems, giving consistent operations everywhere we operate.
Smartway is required to adhere to the strictest of requirements across all jurisdictions we operate in. Whilst we comply with all relevant local regulation, we also ensure that all our sites comply to the highest level of global regulation – adopting the best practices from across the globe and applying it to all our regulated activities. This consistency in our approach means that wherever our clients work with us, they can be assured of consistently high standards.


Our in-house relabelling suite enables us to meet all regulatory requirements for EAP relabelling.
The facility is approved by the UK-MHRA and complies with all relevant international regulation.
Smartway manages the whole process internally, with our regulatory and PM teams ensuring accurate and compliant delivery. This process is scoped out during the design and regulatory compliance stage.

We have adopted the relevant UN Sustainable Development Goals. Our focus in transforming lives by empowering access to medicines globally feeds directly into Goal 3 – Good Health and Wellbeing.
As a global distributor of medicines and pharmaceuticals, we focus on the reduction of carbon in our operations. We work in partnership with our clients to develop carbon neutral programs including working in partnership with a charitable project to trees for carbon capture.
We have developed a series of innovative ways to deliver carbon neutral EAP programs for you.
Please take a look at how else we can help you in EAP.